The Mystery of the Shape of Raindrops

The Size and Shape of Raindrops
How many times have you seen a drawing of a raindrop as something that looks like a drip from a faucet? It's a common symbol that can be found everywhere, from children's books to lawn and garden logos. However, those drawings are perpetuating a very common misconception, that raindrops are shaped like a teardrop.

The way teardrops and faucet drips form is very different from how raindrops form, though some of the molecular principles are the same. Teardrops and faucet drips often fall from a short distance and they sometimes drizzle down a surface. Raindrops form high up in the clouds, under pressure from air, changing temperatures, cooling, and gravity.
Let's take a short cloud journey to discover the shape of raindrops. You know that the air is filled with water vapor, which is essentially a lot of water in gaseous form floating around us. We can't see it, but it's there (hot and humid days demonstrate this quite well). As the Earth is warmed the water vapor rises on a mass of warm air that lifts skyward. The water vapor is warm and bouncing around quite happily, but as the water molecules rise into the the water vapor begins to cool in the atmosphere. Cool air can't hold as much moisture as warm air, there just isn't enough energy, and the vapor becomes sluggish and heavy. When this happens the water molecules in the vapor start to condense. Now they can't just square dance together as water molecules and "ta-da" you have a raindrop, nope, you have to have dust or particulates for them to form around. These are called nuclei, or condensed cloud nuclei (CCN). Nuclei can be anything from pollen or ash to fine grained sand. They're about 1/100 of a millimeter in size, very tiny wee things.

As the water molecules start to condense and cool around the nuclei they begin to form a water droplet that continues to floats along in the atmosphere. The average size of these is about 2 mm. These droplets are perfectly round or spherical, not teardrop shaped. They are round because of the covalent properties of water.
As that raindrop starts to move around, it becomes more heavy and saturated, absorbing other droplets of water. This is sort of like an airborne blob eating up Manhattan. When the drop grows, things get interesting. Any time that this airborne droplet starts to join up with other droplets, and it gets bigger than 2 mm, then it starts to take another shape. This is because the drop is heavier and it begins to be influenced more by air pressure pushing up as the droplet begins to sink down. The droplet forms into something that looks like a jellyfish bell without the tentacles, and it has a slight dimple in the middle of its underside. This droplet will keep descending downward, flattening somewhat. As it goes down it becomes more and more convex (like a contact lens or thin plastic bag). Eventually, when it reaches 4.5-5 mm in size the cohesive molecular bonds of the water molecules can't hold the droplet together anymore, and the droplet simply bursts into smaller round droplets.
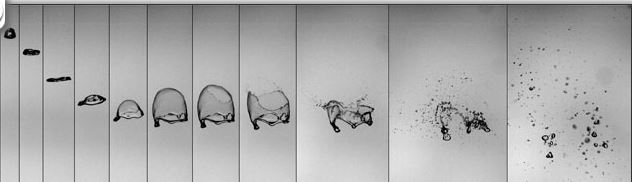
There is some fun geeky weather stuff that goes with all this, especially concerning the amount of air pressure around the large droplet. It has to do with the falling droplet forming a high pressure of air below it as it falls, which creates a low pressure of turbulent air on top of it.
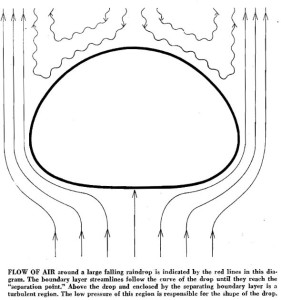
This air pressure differential is what gives the larger droplet its jellyfish shape. If you want to know more you can read the paper by Emmanuel Vilermaux in the Journal Nature.
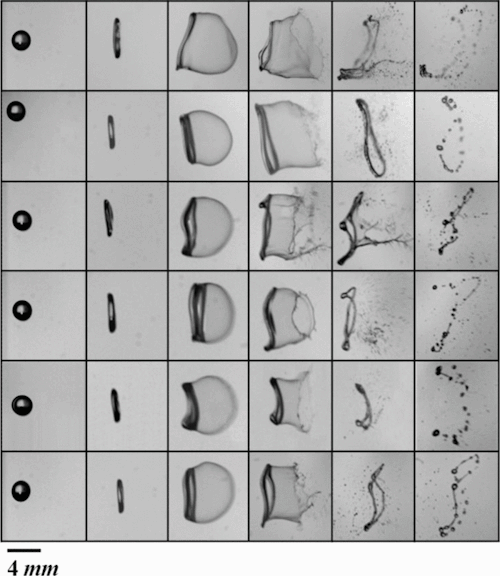
Of course a lot of interesting science went into learning about raindrop shape. The advent of the high speed camera has allowed us to really study fluid movement at such high rates of descent. But my favorite story is that the very first person go get the idea of studying raindrops and their size distribution (which might lead to shape) before high speed camera. This person was better known for studying snowflakes, and is featured in a children's book called "Snowflake Bentley."His name was Wilson Alwyn Bentley, and he lived during the years after the Civil War. He was one of the first to take pictures of snowflakes, and he was fascinated with rain and precipitation in all its shapes and forms.

To study raindrops Bentley would put out a plate of flour and then measure the diameter, distribution, and number of different size raindrops. How did this work? He measured out about 1" of flour, and then exposed it to rain for several seconds. The droplets were suspended and captured in the flour. He then transferred these to paper and when they dried he was able to measure the diameter of the droplets. You can still do this today if you like!
Regardless of the study technique, from flour to high speed photography, we're still learning more and more about the unique properties of water and the shape of raindrops, there is also still much to learn.
Want some fun activities around this for the kids? Try making a cloud in a bottle with matches, a 2 liter bottle, and water. You'll love the snazzy music of this video:
You can also test the covalent properties of water by using a dropper and seeing how many drops fit on the head of a penny. My record is 26!